Talk to us!
Book a complimentary call to get an expert opinion on where you are on your compliance journey.

Squirrel™
The only compliance platform
that is Automated & Assured.





• Trusted by the World's Leading Healthtech Innovators •










































Meet
Squirrel™
We’ve retained the features that made us as smart as a consulting company, as affordable as a compliance platform and better than both... but we’ve also added:

AI-Powered Compliance Copilot
Our new AI compliance assistant is there to help you every step of the way. It uses Leading AI combined with Acorn’s proprietary, compliance-specific knowledge base stored in a secure vector database — delivering precise, context-aware guidance with no hallucinations and full traceability.
Guided by AI, Grounded in Expertise

Squirrel™ helps you draft, structure, and update documentation the way auditors expect, from policies to DPIAs. Every output is backed by expert review.

Ask any question and get real, framework-specific guidance. Your Copilot explains what each clause means, what to do next, and how to prove it, demystifying compliance.

Forget the nightmare of hallucinations. Trained on real regulatory expertise, Squirrel™ meaning the support you get is always proven, traceable and context aware.

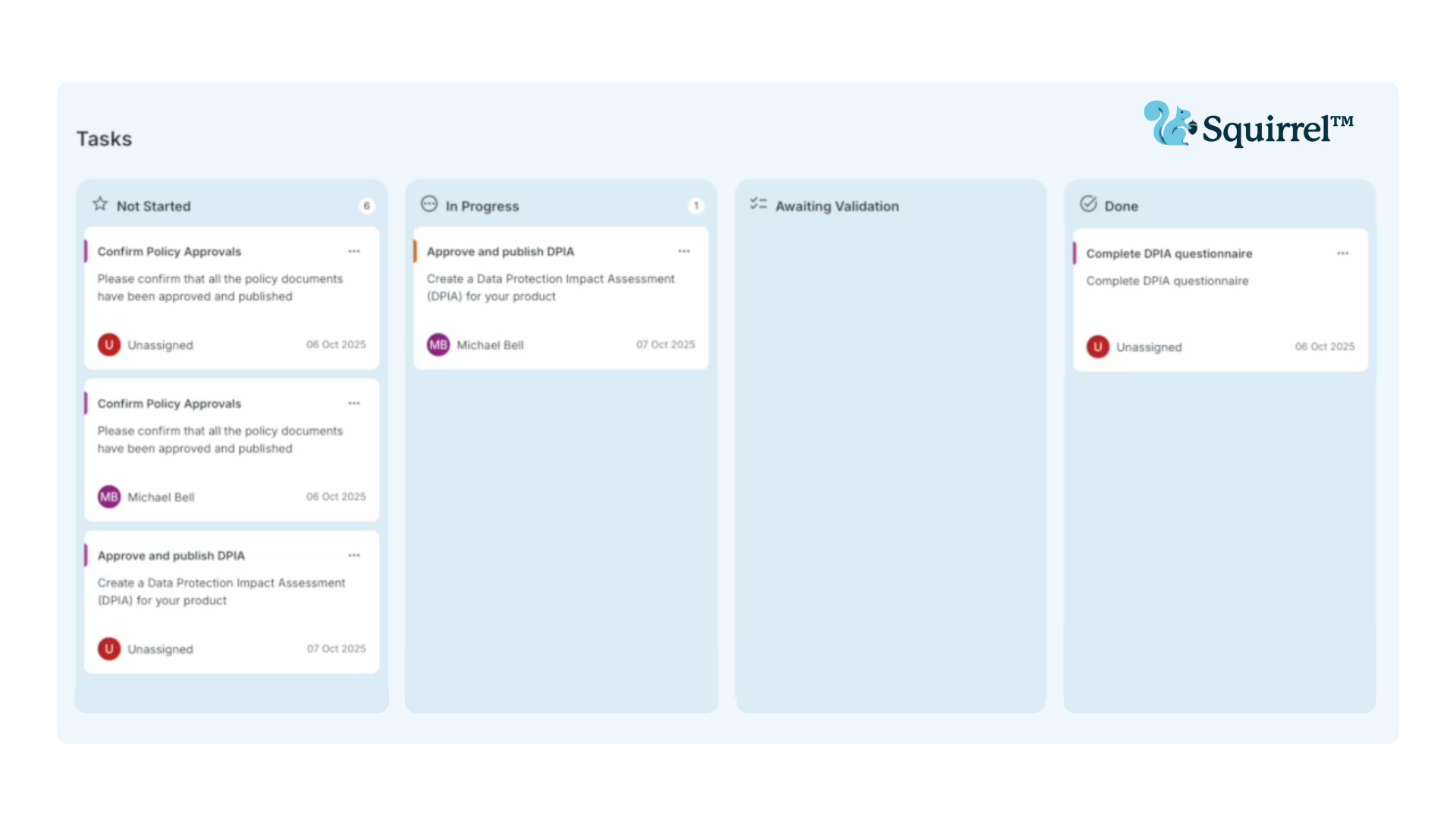
Smart Compliance Planning Dashboard
Turn your compliance roadmap into action with our Kanban-style planning view where tasks are automatically generated with time estimates, helping you plan efficiently and stay on track.


Human-in-the Loop Assurance
Every AI-assisted process is reviewed by compliance professionals and backed by our compliance warranty - ensuring accuracy, accountability, and complete confidence in your compliance outputs.

Unified & Custom Framework Builder
Squirrel™ replaces the need for multiple systems with an all-in-one platform
Adapt frameworks to your organisation, map shared controls automatically, and manage everything from onboarding to audit in one connected workflow.

Ever Evolving Evidence Stack
Regulations evolve, frameworks expand, and evidence requirements shift, especially when you’re managing several at once.
Squirrel™ evolves with you; updating your evidence stack, mapping shared controls, and maintaining continuous compliance across DTAC, ISO, SOC 2, HIPAA, and beyond.
Compliance matters 100% of the time.
Your system should go 100% of the way.
Support that goes (not 20%, not 50%, not even 80%) 100% of the way.
Built For Health
In healthcare, compliance makes or breaks adoption. From first steps through to multi-market scale, Squirrel™ guides you through every compliance framework, DTAC, HIPAA, ISO 27001, MDR and more, in a single automated platform.
End Compliance Silos
Regulated markets demand many frameworks. That shouldn’t mean wasting time and resources doing the same work twice. Squirrel™ centralises tasks and evidence so your effort counts across every standard.
Powerful Automation
Squirrel™ auto-generates context-relevant evidence and keeps it up to date as your product and regulations evolve. Get real-time alerts, collaborate in-platform, and spend less time on compliance admin.
Ready Where You Are
Import all your compliance frameworks, including what you’ve already done. Get a live, prioritised plan, and keep moving without restarting your compliance from scratch.
Expert Guidance, On Demand
Ask your AI copilot for on-demand, cross-framework guidance from within Squirrel™ When it’s time to submit, our compliance experts will review and guarantee your evidence to make sure it holds up to scrutiny.

Squirrel™ is AI-Powered, Expert Backed and Feature Packed
Hundreds of innovators have achieved compliance 10x faster with the confidence that comes from knowing their evidence will hold up to scrutiny. Plus:

More Frameworks

More Markets

More Integration

More Automation

More Efficiency

More Support

Compliance made easy, clients left beaming.
Discover how companies like yours have automated compliance, unlocked new markets, and ditched the compliance chaos.

Save thousands on compliance costs with just one platform.
Compliance can be daunting when you don't see a finish line. Luckily, Squirrel™ can be your friendly guide.
Starter Plan
£249




Standard Plan
£370
Pro Plan
£970
Starter Plan
£2,499




Standard Plan
£3,996
Pro Plan
£10,476
With every package you have the option to purchase add on services as needed:


Squirrel™ Goes Where You Grow

The rest of the world deserves your innovation.
You deserve a platform that won’t let compliance be the bottleneck.

All your frameworks in one place.
From DTAC and ISO 13485 to SOC 2, HIPAA and even your custom frameworks, Squirrel™ brings everything into one platform. Map overlapping controls, manage shared evidence, and stay aligned everywhere you operate.

Cross-Border Compliance, Built In
Expanding into new markets shouldn’t mean starting over. Squirrel™ helps you adapt documentation and processes for each region’s requirements, keeping your global compliance posture consistent across geographies.

Expert Oversight, Wherever You Operate
Our regulatory specialists work alongside your team to review files, validate documentation, and interpret regional standards, ensuring your compliance meets expectations in every market you enter.
Have questions?
I don’t even know where to start!
I only need help with Clinical Safety and DCB 0129. Is that covered?
Is a 12 month contract mandatory?
My product is a medical device — can Squirrel™2.0 help me?
What if I already have some compliance work done?
How does the AI Compliance Copilot actually work?
Can I manage multiple frameworks at once?
Is Squirrel™ 2.0 suitable for organisations outside the UK?
What kind of support will I get?
What if regulations change while I’m mid-project?
Can I add more frameworks or markets later?
You could get compliance assistance from a number of places.
Compliance consultancies
Will reduce some barriers to market entry and
provide guidance for compliance guidance...
But
Might not be very start-up friendly, and will definitely cost an arm and a leg.
Other compliance platforms
Will simplify compliance requirements and save you some time...
But
Might not underwrite your compliance evidence to guarantee that you will pass external audits and assessments.
Reasons Squirrel™ is the best of both worlds:
As smart as a consulting company. As affordable as a compliance platform. Better than both.


And we provide a warranty for all of your evidence, meaning...
We’ve got super-helpful resources
to guide you through compliance.






.png)






















